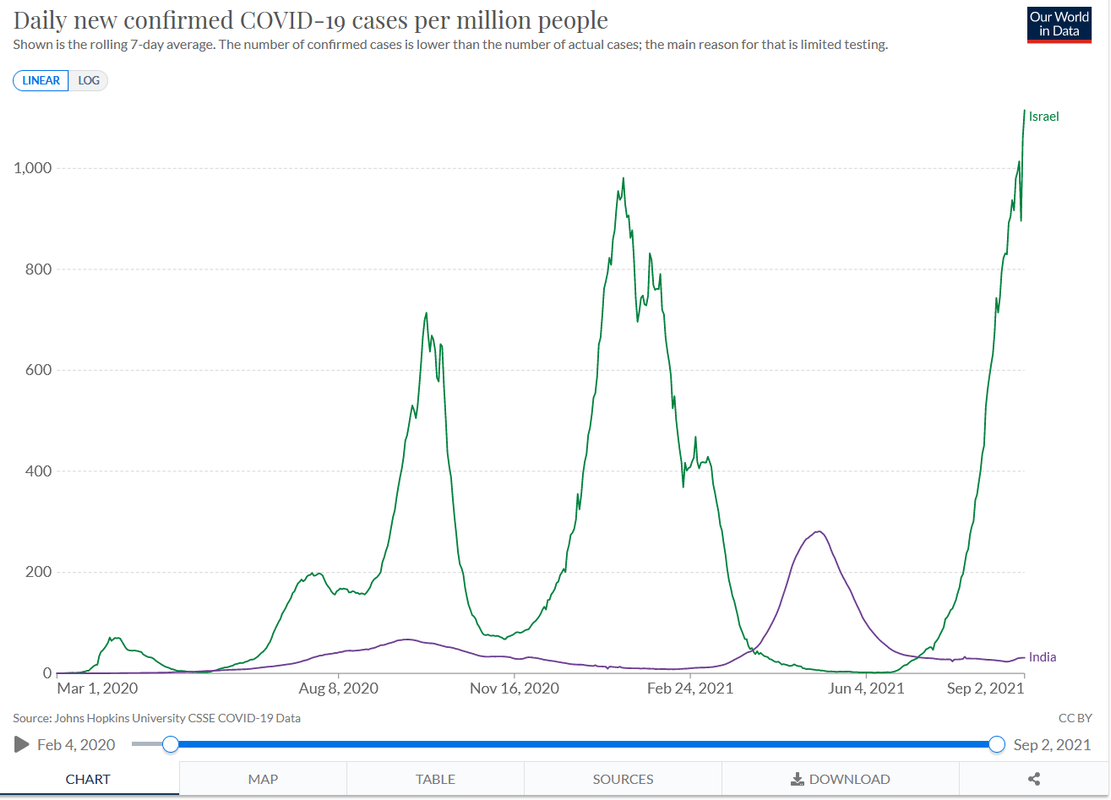
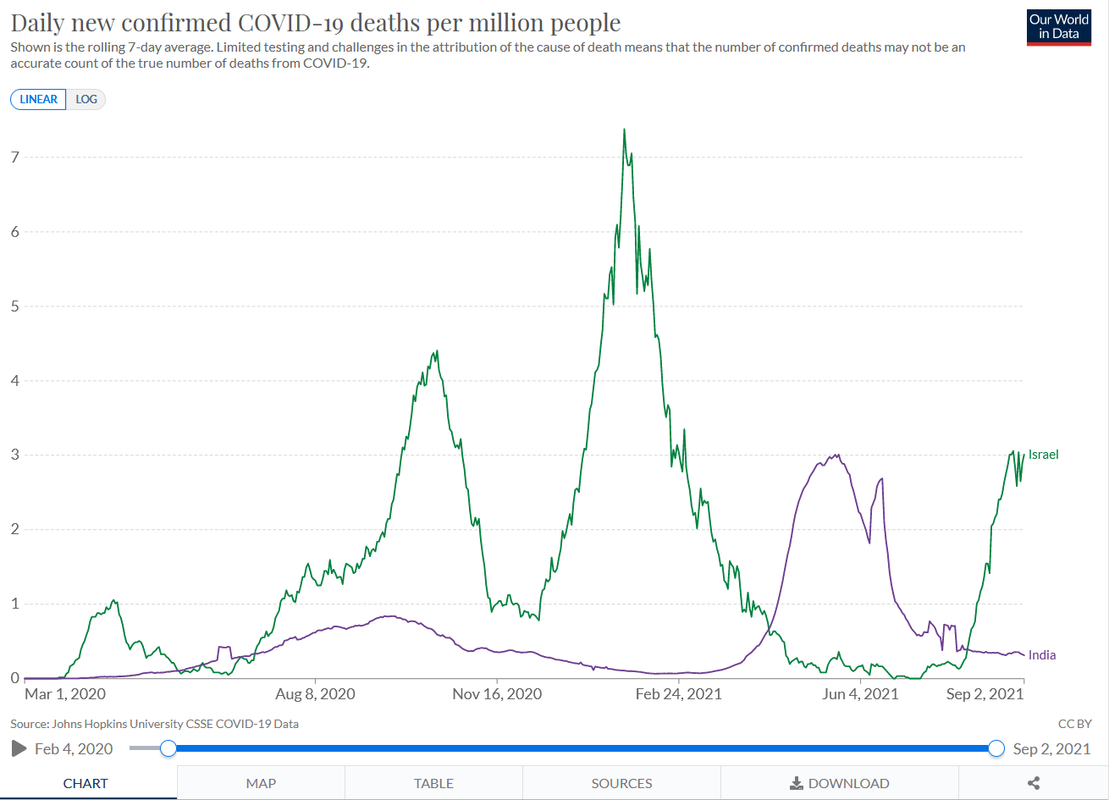
As expected, early results look good.
Now we see how long this round lasts, and what the side effect profile looks like down the road a bit.
https://www.medrxiv....8.27.21262679v1
1,144,690 individuals aged 60y and older who were eligible for a booster dose were followed between July 30 and August 22, 2021. We defined dynamic cohorts where individuals initially belong to the 'non-booster' cohort, leave it when receiving the booster dose and join the 'booster' cohort 12 days later. Rates of infection and severe COVID-19 outcomes per person-days at risk were compared between the cohorts using Poisson regression, adjusting for possible confounding factors. Results: Twelve days or more after the booster dose we found an 11.4-fold (95% CI: [10.0, 12.9]) decrease in the relative risk of confirmed infection, and a >10-fold decrease in the relative risk of severe illness. Under a conservative sensitivity analysis, we find ≈5-fold protection against confirmed infection. Conclusions: In conjunction with safety reports, this study demonstrates the effectiveness of a third vaccine dose in both reducing transmission and severe disease and indicates the great potential of curtailing the Delta variant resurgence by administering booster shots.